 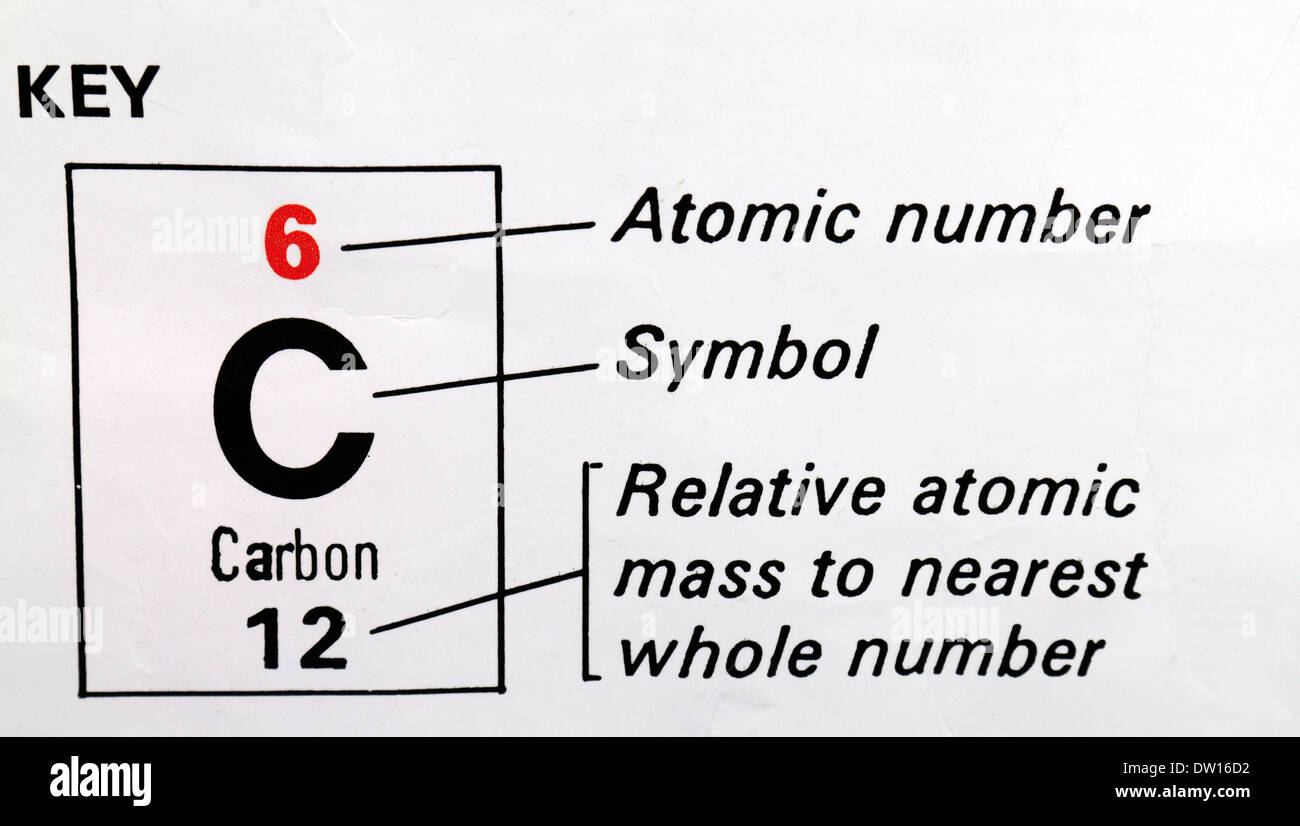
Thus, the mass of the hydrogen atom (1 H) is 1.0080 amu, and the mass of an oxygen atom (16 O) is 15. For example, any atom that contains six protons is the element carbon and has the atomic number 6, regardless of how many neutrons or electrons it may have. The atomic weight of chlorine (35.45) reflects the fact that 75.53. Isotopic masses can be measured using mass spectroscopy. The atomic weight of carbon (12.01) reflects the relative abundance of these three isotopes. The atomic mass unit (amu) was not standardized against hydrogen, but rather, against the 12 C isotope of carbon (amu 12). isotopic mass of carbon-13 atom: 13 nucleons × 1 u/ nucleon 13 u. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. As we saw earlier, it is convenient to use a reference unit when dealing with such small numbers: the atomic mass unit. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. The relative atomic mass of carbon-12 is defined as exactly 12 and the relative atomic mass of carbon-13 is 13.00335. The lightest chemical element is Hydrogen and the heaviest is Hassium. It also compares the isotope abundances of carbon in the atmosphere and in fossils. The web page explains the properties of atoms, the mass number, the relative atomic mass, and the half-life of radioactive isotopes of carbon. Other atoms typically do not have round-number atomic masses. Learn how to calculate the atomic mass of carbon and its isotopes, and how to distinguish between stable and radioactive forms of carbon. Carbon-12, an atom of carbon-containing six neutrons, has an atomic mass of 12 amu. A single atom’s atomic mass is just its total mass, and it is commonly given in atomic mass units or amu. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). The atomic mass of an atom is a feature that is closely related to its mass number. Only one out of every trillion carbon atoms is 14C. There is an alternative phrasing for this which some. While only approximately 1 of all carbon on Earth is of the 13C isotopic form, 14C is still much rarer. 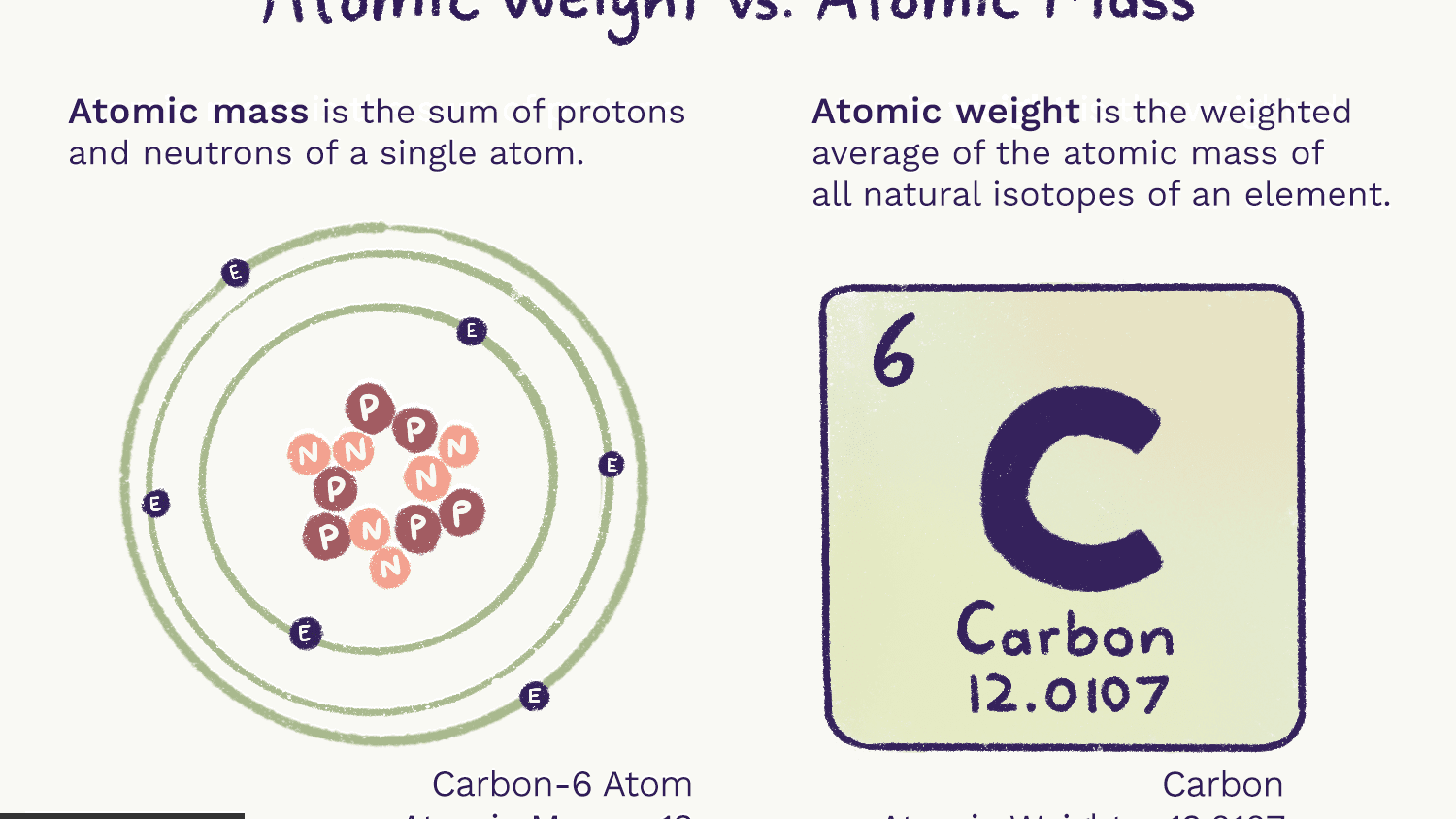
This list contains the 118 elements of chemistry. The relative isotopic mass is the mass of the isotope relative to 1/12 of the mass of a carbon-12 atom. Average masses are generally expressed in unified atomic mass units (u), where 1 u is equal to exactly one-twelfth the mass of a neutral atom of carbon-12. Its presence in organic materials is the basis of the radiocarbon dating method pioneered by Willard Libby and colleagues (1949) to date archaeological, geological and hydrogeological samples. The average atomic mass (sometimes called atomic weight) of an element is the weighted average mass of the atoms in a naturally occurring sample of the element. 
Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects. C or radiocarbon, is a radioactive isotope of carbon with an atomic nucleus containing 6 protons and 8 neutrons. Experimentally, the weight of Avogadros number (NA 6.023 × 1023) of carbon atoms, each containing six protons and six neutrons, equals 12.00000 g, where.The difference between the neutron number and the atomic number is known as the neutron excess: D. Neutron number plus atomic number equals atomic mass number: N+ZA. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. Plant Inspection & Process Optimalisation Mass numbers of typical isotopes of Carbon are 12 13.Since Avogadro's number is a measured quantity with three significant figures, the result of the calculation is rounded to three significant figures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
